Mesenchymal stem cell research: Current trends and attitudes in
RegMedNet recently conducted a survey regarding the current trends and future developments in mesenchymal stem cell research. The survey was available to all members of the RegMedNet community and was completed by scientists, students, technicians and group leaders from both academia and from biotechnology and medical device companies. The survey collected information on the type of MSC research currently being undertaken, the type and source of stem cells used, what conditions the MSCs are cultured in and the current challenges facing researchers working with MSCs amongst other things.
The survey showed that adipose-derived MSCs are the most commonly utilised mesenchymal stem cells, closely followed by MSCs sourced from bone marrow. Regardless of the source of the MSCs, 90% of researchers that took part in the survey are utilising MSCs for regenerative medicine purposes. This further emphasises the fact that MSCs have become a very attractive and desirable tool in tissue engineering and cell therapy approaches. Only 14% were using MSCs for disease modelling and 11% for drug discovery and cytotoxicity testing.
The survey appears to suggest that musculoskeletal disease states and conditions, including arthritis, are the area that are attracting the most studies into the therapeutic use of MSCs. This may be unsurprising due to their multi-lineage differentiation potential and well-documented differentiation into chondrocytes, osteocytes and adipocytes. The disease states and indications that MSCs are being employed in vary greatly though. This is highlighted in the fact that there are multiple studies on-going into the causes and treatment of spinal cord injuries, cardiovascular diseases and critical limb ischemia, to name just a few.
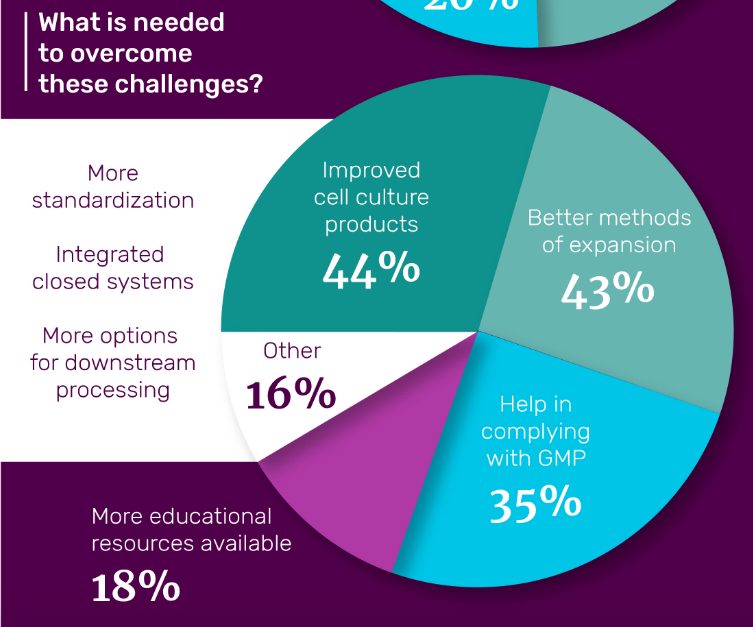
One of the statistics to come out of this survey that was particularly interesting was relating to the challenges that those undertaking mesenchymal stem cell research face and what is needed to overcome them. Around a quarter of those surveyed expressed their biggest problem being with maintaining the phenotype and function of their MSCs and a fifth of people stated the biggest issue involved the expansion of the cells. 44% of people have specified improved cell culture products as the thing that is needed to overcome these issues.
To any researchers experiencing these problems I would strongly recommend trying Biological Industries MSC Nutristem media and requesting a sample from us. The quality of the culture medium that you use and its performance are especially important with regards to the therapeutic applications, since the properties of hMSCs are significantly affected by medium components and culture conditions. By using MSC Nutristem you are using a defined, serum-free and xeno-free medium optimised for the isolation and expansion of hMSCs, from a variety of sources (AT, WJ, BM, DP, PL, UC), which facilitates the development of robust, clinically acceptable culture processes for reproducibly generating quality-assured cells.
To keep up to date with more news and information, on mesenchymal stem cell research and much more, make sure to follow us on Twitter: @CellsecoLtd