MSC Nutristem XF Medium
Defined, xeno-free, serum-free MSC culture medium
Eliminates the need to spend time and money prequalifying FBS lots
GMP-defined, FDA Drug Master File (DMF) available
| name | SKU | size |
|---|---|---|
| MSC NutriStem® XF Medium (Basal Medium + Supplement) (US only) | 05-200-1A-KT | 500 mL |
| MSC NutriStem® XF Basal Medium | 05-200-1A | 500 mL |
| MSC NutriStem® XF Basal Medium, without Phenol Red | 05-202-1A | 500 mL |
| MSC NutriStem® XF Supplement Mix | 05-201-1U | 3 mL |
- Description
- Specifications
- References
- Documentation
- Reviews (0)
Description
MSC Nutristem Overview:
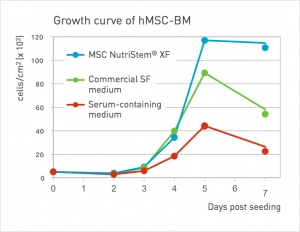
Figure: MSC NutriStem XF Medium promotes superior proliferation and expansion of hMSCs over time as compared to other serum-free and serum-containing media.
Faster growth rates and stronger MSC yields
The quality of the culture medium used for expansion of hMSCs is crucial to the potential therapeutic applications of the cells, since hMSCs properties are significantly affected by medium composition and culture conditions.
MSC Nutristem XF Medium is a defined, serum-free, xeno-free culture medium specially designed to support the growth of hMSC derived from a variety of sources that include: bone marrow (hMSC-BM), adipose-tissue (hMSC-AT), Wharton’s Jelly (hMSC-WJ), placenta (hMSC-PL), umbilical-cord matrix (hMSC-UC) and dental pulp (hMSC-DP).
Key advantages
- Serum-free, xeno-free, defined culture system
- Superior cell growth compared to serum-containing media
- Supports long-term growth and self-renewal of hMSCs from multiple sources
- cGMP-manufactured, FDA Drug Master File (DMF) available
- Superior maintenance of hMSC characteristics and cell morphology
- Karyotypic stability and over long-term culture
- Customisation available and customisable packaging configurations (i.e 1-20L bags)
Cells cultured in serum-containing medium do not require an adaptation phase when transitioning to serum-free MSC NutriStem XF Medium.
Sample MSC Nutristem Data
Initial isolation
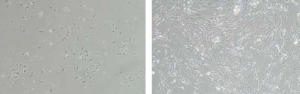
Figure: Human mesenchymal stem cells were initially isolated from Wharton’s jelly (left image) and placenta (right image) using MSC NutriStem XF on pre-coated plates with MSC Attachment Solution (cat. no. 05-752-1). High number of hMSC was obtained.
Proliferation
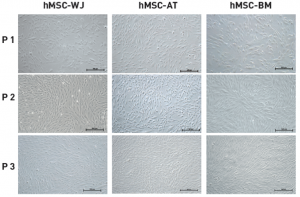
Figure: MSC NutriStem® XF promotes excellent proliferation of hMSC from a variety of sources while maintaining normal fibroblast-like, spindle shape cell morphology.
Self-renewal potential
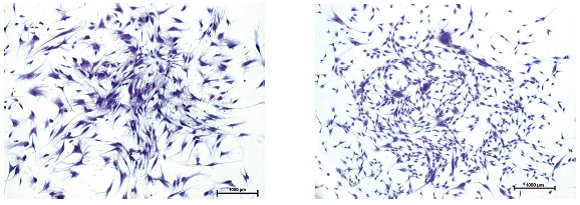
Figure: hMSC-BM and hMSC-AT were expanded in MSC NutriStem® XF for 3-5 passages prior to 14 days of CFU-F assay. Colonies were stained with 0.5% crystal violet (x100). The cells maintain their self-renewal potential
Differentiation potential
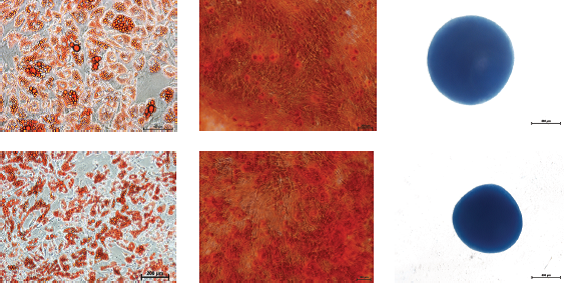
Figure: hMSC-BM (top panel) and hMSC-AT (bottom panel) expanded in MSC NutriStem® XF for 3-5 passages prior to trilineage differentiation. Representative images of stained adipocytes (Oil Red O), osteocytes (Alizarin Red) and chondrocytes (Alcian Blue). The cells maintain their multilineage differentiation potential
Instructions for Use
MSC NutriStem® XF Medium is recommended for use in conjunction with MSC Attachment Solution (Cat. No. 05-752-1).
Storage and Stability
- MSC NutriStem® XF Basal Medium (05-200-1) should be stored at 2 to 8°C.
- MSC NutriStem® XF Supplement Mix (05-201-1) should be stored at -20°C. Avoid freeze/thaw cycles.
- The complete MSC NutriStem® XF Medium is stable at 2 to 8°C for up to 30 days.
- Avoid exposure to light
Medium Preparation
- Thaw frozen MSC NutriStem® XF Supplement Mix at at 2 to 8°C or room temperature
- MSC NutriStem® XF Basal Medium contains L-glutmine
- To prepare 100 mL of complete medium: aseptically add 0.6 mL of MSC NutriStem® XF Supplement Mix to 100 mL of MSC NutriStem XF Basal Medium
- To prepare 500 mL of complete medium: aseptically add 3 mL of MSC NutriStem® XF Supplement Mix to 500 mL of MSC NutriStem XF Basal Medium
- Store complete MSC NutriStem® XF Medium at 2 to 8°C protected from light for up to 30 days
Additional information
| Quantity | 3 mL, 500 mL |
|---|---|
| Brand | |
| Form | Liquid |
- C. Elseberg et al. The Challenge of Human Mesenchymal Stromal Cell Expansion: Current and Prospective Answers. New Insights into Cell Culture Technology, Dr. Sivakumar Joghi Thatha Gowder (Ed.), InTech.
- C. Ceccaldi et al. Optimization of Injectable Thermosensitive Scaffolds with Enhanced Mechanical Properties for Cell Therapy. Macromolecular Bioscience, 2017
- D. Boruczkowski et al.Third-party Wharton’s jelly mesenchymal stem cells for treatment of steroid-resistant acute and chronic graft-versus-host disease: a report of 10 cases. Turkish Journal of Biology, 40: 493-500, 2016
- Jianxia H. et al. Long term effect and safety of Wharton's jelly-derived mesenchymal stem cells on type 2 diabetes. Experimental and Therapeutic Medicine, Volume 12 Issue 3, 2016
- S. Bobis-Wozowicz et al. Diverse impact of xeno-free conditions on biological and regenerative properties of hUC-MSCs and their extracellular vesicles. Journal of Molecular Medicine, 2016
- L. Berger et al. Tumor Specific Recruitment and Reprogramming of Mesenchymal Stem Cells in TumorigenesisSTEM CELLS Volume 34, Issue 4, Version of Record online: 31 DEC 2015
- K.Y. Tan et al. Serum-free media formulations are cell line–specific and require optimization for microcarrier culture. Cytotherapy, 2015
- Cai, Zhen, et al. Chondrogenesis of Human Adipose-Derived Stem Cells by In Vivo Co-graft with Auricular Chondrocytes from Microtia. Aesthetic plastic surgery 39.3 (2015): 431-439.
- A. Jarmalavičiūtė et al., Exosomes from dental pulp stem cells rescue human dopaminergic neurons from 6-hydroxy-dopamine–induced apoptosis. Cytotherapy, 2015
- M.Pokrywczynska et al., Transdifferentiation of Bone Marrow Mesenchymal Stem Cells into the Islet-Like Cells: the Role of Extracellular Matrix Proteins. Archivum Immunologiae et Therapiae Experimentalis, May 2015
- U Pivoraitė et. al., Exosomes from Human Dental Pulp Stem Cells Suppress Carrageenan-Induced Acute Inflammation in Mice. Inflammation, April 2015
- S.H. Mei, et al. Isolation and large-scale expansion of bone marrow-derived mesenchymal stem cells with serum-free media under GMP-compliance. Cytotherapy, Volume 16, Issue 4, Supplement , Page S111, April 2014
- Mira Genser-Nir et al. Toward a serum-free, xeno-free culture system for optimal growth and expansion of hMSC suited to therapeutic applications. From 23rd European Society for Animal Cell Technology (ESACT), Meeting: Better Cells for Better Health.Lille, France. 23-26 June 2013
- McVey, Mark John, et al. Microparticles as biomarkers of lung disease-enumeration in biological fluids using lipid bilayer microspheres. American Journal of Physiology-Lung Cellular and Molecular Physiology (2016): ajplung-00369.
- Y. Lopez, et al. Identification of Optimal Conditions For Generating MSCs For Preclinical Testing: Comparison of Three Commercial Serum-Free Media And Low-Serum Growth Medium. From 18th ISCT Annual Meeting, Seattle, USA, 2012.
- R. D. Foster et al. GorodetskyFibrin microbeads loaded with mesenchymal cells support their long-term survival while sealed at room temperature.Tissue Eng Part C Methods2011TISSUE ENGINEERING: Part C, Volume 17, Number 7, 2011
Materials Safety Data Sheet
Manuals and Protocols
Product Literature
MSC NutriStem® XF Medium Product Sheet
Blog Posts & Articles
- Blog: Top 5 Reasons to Use NutriStem® for Isolation and Growth of hMSCs (BioInformant)
- Blog: Facilitating the Use of MSCs for Regenerative Medicine Applications through Serum-Free, Xeno-Free Differentiation Media (BioInformant)
Frequently Asked Questions
Be the first to review “MSC Nutristem XF Medium”
You must be logged in to post a review.










Reviews
There are no reviews yet.